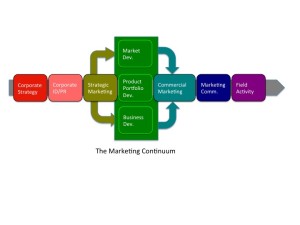
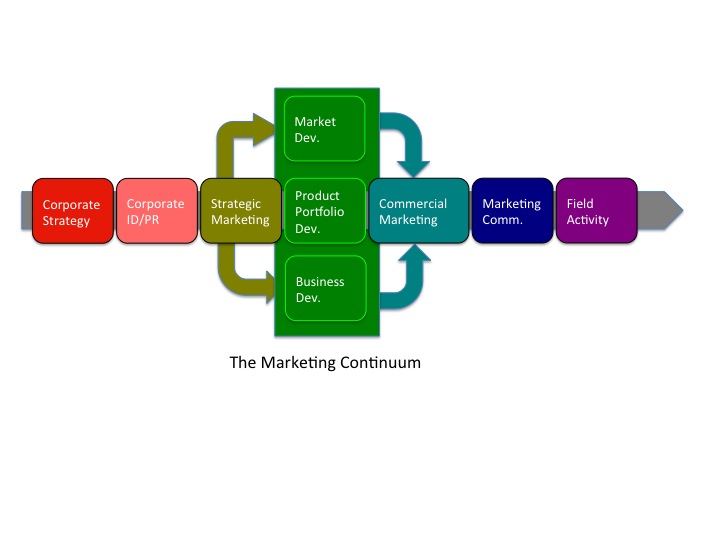
At the heart of the Marketing continuum are three aspects of strategic marketing: 1) New Product Portfolio Development, 2) Market Development, and 3) New Business Development.
Are they really different? Yes and No is the answer. To be successful in any of the three areas you need a strong understanding of the core principles of marketing, a great understanding of your organizations capabilities and of course, a strong understanding of the nature of the customer and the environment in which they work.
The analytic tools you use will be very similar. The basics of great messaging will apply. Where they differ is in the nature of the problem you are trying to solve.
Market Development Defined
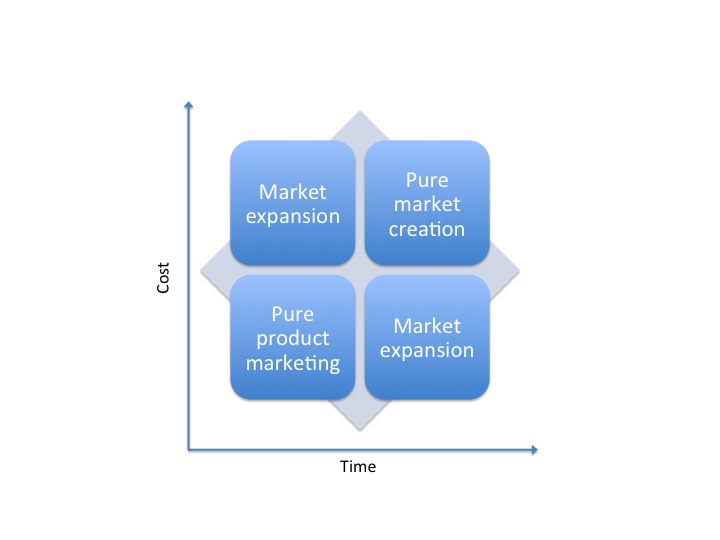
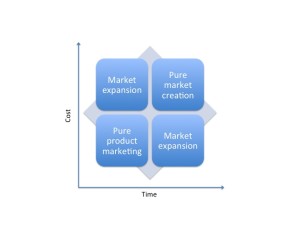
Market Development is simply the creation or expansion of a market. To expand a market or create a market you have to first “sell” the idea that a problem exists. You need to educate the potential buyers that they have an un-met need that they were unaware of.
Product Marketing Defined
With Product Marketing you are “selling” the solution to an already established problem or un-met need.
The Question is, “do you ever have to do both at the same time?”
The answer is yes, to varying degrees.
Typically, it is very expensive and a slow process to develop a market from scratch. There are many benefits in being the leader who creates a market. Typically the first mover advantage will provide leverage in the market place right up until someone develops a better solution.
On a relative scale product marketing is quicker and less expensive than creating markets.
The Story
A client of mine has a great product. There is a real clinical problem that this product solves. There are three or four “use cases” for this product. Some of the use cases are obvious to the key stakeholders, some aren’t. The strategic marketing challenge is where to place the available funds? Which will drive the right kind of success?
It is not always obvious what to do. What will bring the most success for the least investment? It is times like these, when you are facing complex strategic questions when I fall back on the core principles and tools of marketing.
- When in doubt ask a customer (s):
- Who is/are the buyer(s)?
- Who are the key none buying influencers?
- Are the problem(s) that you are solving the same or different?
- Is the product the right product for all use cases?
- What are the barriers to success?
- Is there a genuine value proposition for all stakeholders?
- Is the value proposition strong enough to make it worth the users time to be educated?
- What resonates with the customers?
- What evidence or proof will the buyer need to accept your proposition?
- Scan the environment:
- How large is each use case opportunity?
- Is there competition or are you substituting an alternative solution?
- Is there new technology on the horizon?
- New laws or regulations that are coming or that are needed to provide leverage?
- Are there any parrallel examples of successful strategies
- Craft a hypothesis strategy:
- Test your hypothesis
- Model the potential results of your strategy
- Select a strategy
- Fire a bullet not a cannon ball[1]
There are no formulae for crafting great market development strategies. You have to eliminate the non-starters and then design tests to explore the ones you have hope for.
“Experience is what you get, right after you need it most.”
Make it a great day,
Tim Walker
Tim Walker is the Principal consultant for The Experia Group. A small consulting firm that specializes in providing experience and expertise during critical device commercialization phases to increase the probability of success. www.theexperiagroup.com.
© 2015 The Experia Group, LLC
[1] Great by Choice, Jim Collins, Harper Business Press, Chapter 4, p. 60-98.